What is a fuel cell
How does it work?
The concept of a fuel cell is not new. It has been demonstrated in the XVIIIth century by the scientists Christian Friedrich Schönbein and William Grove – who is generally credited with inventing the fuel cell in 1839. The term “fuel cell” was firstly used in 1889.
Grove conducted a series of experiments and proved that electric current could be produced from an electrochemical reaction between hydrogen and oxygen over a platinum catalyst.
The fuel cell concept is simple :
Hydrogen + Oxygen → Electricity + Water + Heat
2 H2 + O2 → 2 H2O
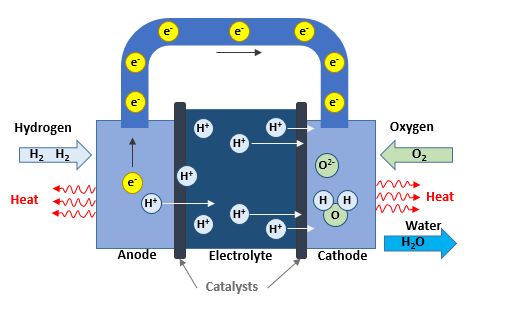
Inside the fuel cell, a redox reaction takes place to produce electricity and heat. For a PEM Fuel Cell, Hydrogen is processed at the anode where electrons are separated from protons on the contact of a catalyst. It is the oxidation step.
2 H2 → 4 H+ + 4 e–
The Hydrogen protons migrate through a membrane, then cross the electrolyte to reach the cathode side. As electronic insulator, the membrane stops electrons and they have to reach the cathode side through an external circuit.
On the cathode side, Oxygen is carried out (extracted directly from the air) and is combined with electrons on the surface of a catalyst. This is the reduction step.
O2 + 4 e– → 2 O-2
Finally, the Hydrogen protons are combined with Oxygen anions to produce water.
4 H+ + 2 O-2 → 2 H2O
Even if the concept seems to be simple, in reality it is more complex to develop a highly-efficient and durable fuel cell.
When were fuel cells developed ?
The first fuel cell was developed in the late XIXth century but the technology remained obscure until Francis T. Bacon – an engineering professor at Cambridge University – developed an Alkaline Fuel Cell (AFC) in the late 1930s and demonstrated the first fuel cell system with a nominal power of 5 kW in 1959.
Research activities led by NASA with industrial partners improved fuel cell lifespan and fuel cell generators were used for space missions.
Nevertheless, the technology still remained too expensive. The keen interest for fuel cell systems and for hydrogen technologies reemerged with concerns about climate change and energy security. The automotive industry launched a huge research program in the early 1990s and several fuel cell systems for automotive purpose were developed.
What are the different fuel cell technologies ?
We can identify :